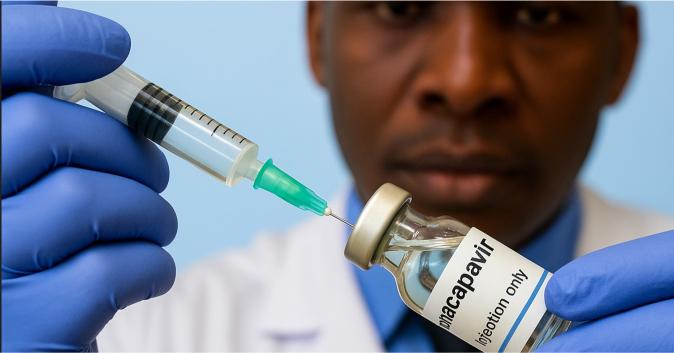
Uganda has given the green light for the use of Lenacapavir, a long-acting HIV prevention injection administered twice a year, marking a significant advance in the country’s efforts to curb HIV/AIDS.
The injection, developed by Gilead Sciences in the United States, offers a practical alternative for people who find daily pills challenging to maintain or face stigma when accessing healthcare services.
Lenacapavir, which received World Health Organization (WHO) endorsement in July 2025 and prequalification in October, delivers protection with just two shots per year. Uganda’s National Drug Authority described the approval as a “game changer” in a nation where roughly 5.4 per cent of adults live with HIV.
“This is a game-changer for HIV prevention, especially for those at high risk. This is a great step towards ending AIDS by 2030,” the statement said.
With this move, Uganda joins South Africa, Zimbabwe, and Zambia in offering Lenacapavir on the continent. WHO noted that other countries are pursuing approval under its Collaborative Registration Procedure, which aims to broaden access to the long-acting injection across Africa.
In Kenya, authorities plan to introduce the drug later this year, although the current cost remains a concern. Each dose is expected to sell for about Sh6,000, but partnerships with international organizations and generic manufacturers could lower the price for the two yearly injections to Sh5,000–5,200. Health officials hope generics will eventually make each injection available at Sh2,500–2,600.
Kenya continues to grapple with a substantial HIV burden, with approximately 1.33 million people living with the virus. National prevalence stands at 3.0 per cent, affecting women more heavily at 4.1 per cent compared to 1.95 per cent among men.
In 2025, the country reported around 20,105 new HIV infections and 21,009 AIDS-related deaths, showing that prevention efforts remain a critical priority.






























Comments
Sign in with Google to comment, reply, and like comments.
Continue with Google